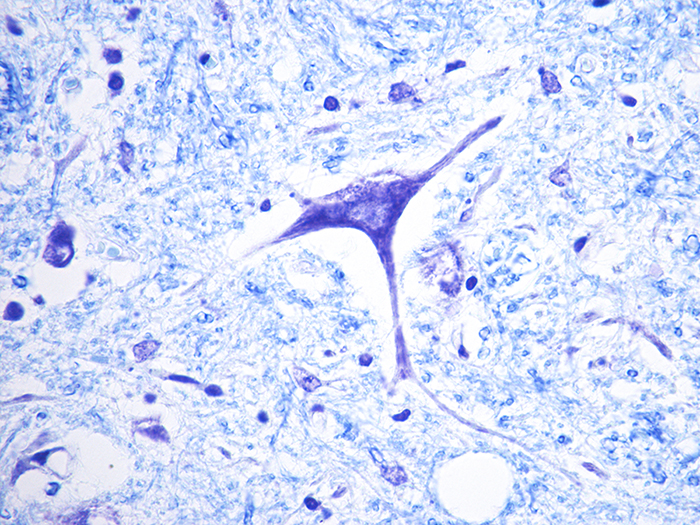
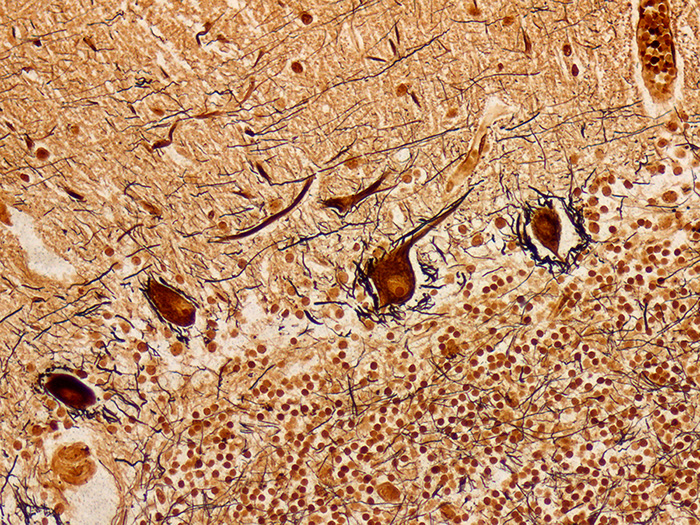
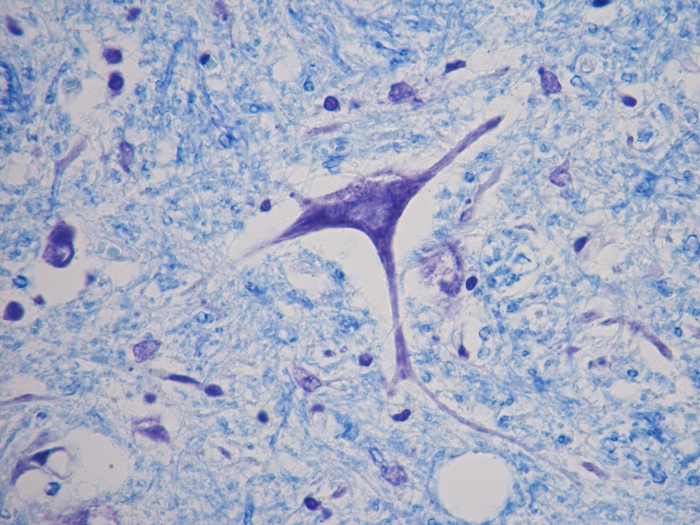
Luxol Fast Blue (LFB)
|
Validation Stain: Luxol Fast Blue (LFB) – Cresyl Violet
Other Applicable Stains: LFB, Bodian, Holzer, PTAH and Weil’s
|
-
- Tissue sections are cut at 8 microns
PRODUCT SPECIFICATIONS:
Tissue: Positive staining spinal cord.
Fixation: Formalin 10%, Phosphate Buffered (Part 1090).
Section/Glass: Paraffin sections cut at 8 microns on Superfrost™ Plus slides.
Quality Control Stain: Luxol Fast Blue quality control stained slide(s) included.
Reactivity: Guaranteed product specific reactivity for one year from date of receipt. Revalidate after one year to verify continued reactivity.
Storage: 15-30°C in a light deprived and humidity controlled environment.
Intended Use: To verify histological techniques and reagent reactivity.
Before using unstained control slides, review the enclosed stained slide(s) to ensure that this tissue source is acceptable for testing needs.
CONTROL SLIDE VALIDATION:
| With Luxol Fast Blue (LFB) Stain Set: | Part 12218A/B | Individual Stain Solution | |
| Solution A: | Luxol Fast Blue Stain 0.1%, Alcoholic | 500/1000 ml | |
| Solution B: | Lithium Carbonate, Saturated Aqueous | 500/1000 ml | Part 12215 |
| Hematoxylin Stain, Harris Modified | Part 1201 | ||
| Acid Alcohol 1% | Part 10011 | ||
APPLICATION:
Newcomer Supply Luxol Fast Blue (LFB) Control Slides are for the positive histochemical staining of myelin sheath in central nervous system tissue and in peripheral nerve. PAS, cresyl violet, hematoxylin and eosin stains can be added to LFB procedure for enhanced staining.
PRESTAINING PREPARATION:
-
- Heat dry sections in oven according to your laboratory protocol.
- Prepare Working Lithium Carbonate 0.05%; combine and mix well;
-
- Solution B: Lithium Carbonate, Saturated Aqueous 5 ml
- Distilled Water 95 ml
-
STAINING PROCEDURE:
-
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each.
-
- Stop at 95% ethyl alcohol; no distilled water rinse.
- See Procedure Notes #1 and #2.
-
- Incubate slides in Solution A: Luxol Fast Blue Stain 0.1%, Alcoholic for 2 hours at 60°C or overnight at 37°C; seal lids tightly.
-
- To enhance stain, add 0.4 ml of Acetic Acid, Glacial, ACS (Part 10010) to 40 ml of Solution A: Luxol Fast Blue Stain 0.1%, Alcoholic before use.
-
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each.
Microwave Modification: See Procedure Note #3.
-
-
-
- Place slides in a plastic Coplin jar with Solution A: Luxol Fast Blue Stain 0.1%, Alcoholic. Microwave at 70°C for 10 minutes.
-
-
-
- Rinse slides quickly in 95% ethyl alcohol; 2-3 dips.
- Rinse slides in distilled water.
- Differentiate slides individually in Working Lithium Carbonate 0.05% (Step #2) for 10-15 seconds with agitation until gray and white matter are colorless and contrast with stained tissue.
- Further differentiate in 70% ethyl alcohol (Part 10844), until gray and white matter can be distinguished. Do not over differentiate.
- Rinse slides in distilled water.
- Check slides microscopically. Continue if additional differentiation is needed. Otherwise proceed directly to Step #12.
-
-
-
- One dip in Lithium Carbonate 0.05%, Aqueous (Step #2).
- Dip in two changes of 70% ethyl alcohol until greenish/blue white matter sharply contrasts with colorless gray matter.
-
-
-
- Rinse thoroughly in distilled water.
- Stain with Hematoxylin Stain, Harris Modified (Part 1201) for 1-5 minutes, depending on preference of intensity.
- Wash in running tap water for 3 minutes.
- Differentiate quickly in Acid Alcohol 1% (Part 10011); 3 dips.
- Wash well in running tap water.
- Blue in Solution B: Lithium Carbonate, Saturated Aqueous.
- Wash well in running tap water.
- Dehydrate in two changes each of 95% and 100% ethyl alcohol. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
RESULTS:
| Myelin | Blue to blue/green |
| Nissl substance and nuclei | Blue |
PROCEDURE NOTES:
-
- Drain slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during procedure.
- The suggested microwave procedure has been tested at Newcomer Supply. This procedure is a guideline and techniques should be developed for use in your laboratory.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps
REFERENCES:
-
- Bancroft, John D., and Marilyn Gamble. Theory and Practice of Histological Techniques. 6th ed. Oxford: Churchill Livingstone Elsevier, 2008. 378.
- Carson, Freida L., and Christa Hladik Cappellano. Histotechnology: A Self-instructional Text. 4th ed. Chicago: ASCP Press, 2015. 206-211.
- Klüver, Heinrich, and Elizabeth Barrera. “A Method for the Combined Staining of Cells and Fibers in the Nervous System.” Journal of Neuropathology and Experimental Neurology4 (1953): 400-403.
- Modifications developed by Newcomer Supply Laboratory.