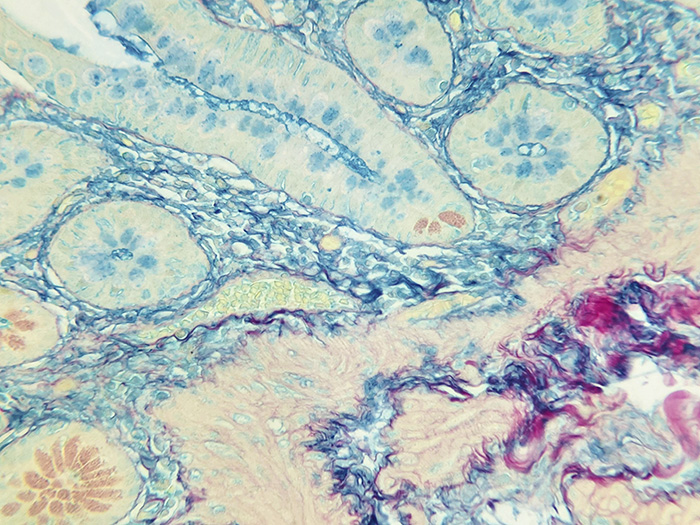
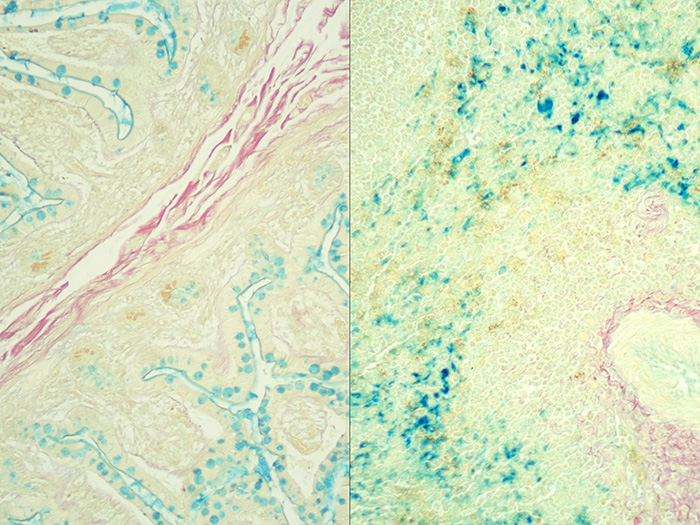
Colloidal Iron, Müller-Mowry Stain Kit
Stains acid epithelial mucin (sialomucin, sulfomucin) and stromal (mesenchymal) mucin and also demonstrates encapsulated yeast Cryptococcus neoformans.
COLLOIDAL IRON, MÜLLER-MOWRY STAIN KIT INCLUDES:
| Part 9110A | ||
| Solution A: | Acetic Acid 12%, Aqueous | 500 ml x 2 |
| Solution B: | Colloidal Iron Stock | 125 ml |
| Solution C: | Acetic Acid, Glacial, ACS | 50 ml |
| Solution D: | Potassium Ferrocyanide 2%, Aqueous | 125 ml |
| Solution E: | Hydrochloric Acid 2%, Aqueous | 125 ml |
| Solution F: | Van Gieson Stain | 250 ml |
COMPLIMENTARY POSITIVE CONTROL SLIDES: Enclosed are two complimentary unstained positive control slides for the initial verification of staining techniques and reagents. Verification must be documented by running one Newcomer Supply complimentary positive control slide along with your current positive control slide for the first run. Retain the second complimentary control slide for further troubleshooting, if needed.
Individual stain solutions and additional control slides may be available for purchase under separate part numbers.
Additionally Needed:
| Hydrochloric Acid 5%, Aqueous | Part 12086 (for acid cleaning glassware) |
| Xylene, ACS | Part 1445 |
| Alcohol, Ethyl Denatured, 100% | Part 10841 |
| Alcohol, Ethyl Denatured, 95% | Part 10842 |
For storage requirements and expiration date refer to individual bottle labels.
APPLICATION:
Newcomer Supply Colloidal Iron, Mϋller-Mowry Stain Kit procedure is used to demonstrate acid epithelial mucin (sialomucin, sulfomucin) and stromal (mesenchymal) mucin. This method is also excellent for the demonstration of the encapsulated yeast Cryptococcus neoformans.
METHOD:
Fixation: Formalin 10%, Phosphate Buffered (Part 1090)
Technique: Paraffin sections cut at 4 microns
Solutions: All solutions are manufactured by Newcomer Supply, Inc.
All Newcomer Supply Stain Kits are designed to be used with Coplin jars filled to 40 ml following the staining procedure provided below. Some solutions in the kit may contain extra volumes.
PRESTAINING PREPARATION:
-
- If necessary, heat dry tissue sections/slides in oven.
- Acid clean glassware prior to use to avoid residual iron staining.
-
- See Procedure Note #1.
-
- Prepare Colloidal Iron Working Solution; combine and mix well.
-
- Solution B: Colloidal Iron Stock 20 ml
- Solution C: Acetic Acid, Glacial, ACS 5 ml
- Distilled Water 15 ml
-
STAINING PROCEDURE:
-
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
-
- See Procedure Notes #2 and #3.
-
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
-
- Place in Solution A: Acetic Acid 12%, Aqueous for 30 seconds.
- Drain Slides. Do not rinse.
- Place in Colloidal Iron Working Solution (Step #3) for 30 minutes.
- Rinse in three changes of Solution A: Acetic Acid 12%, Aqueous; 3 minutes each.
- Prepare fresh Ferrocyanide-Hydrochloric Acid Solution directly before use; combine and mix well.
-
- Solution D: Potassium Ferrocyanide 2%, Aqueous 20 ml
- Solution E: Hydrochloric Acid 2%, Aqueous 20 ml
-
- Place in Ferrocyanide-Hydrochloric Acid Solution for 15 minutes.
- Wash in running tap water for 1-5 minutes.
- Counterstain in Solution F: Van Gieson Stain for 3-5 minutes.
-
- Proceed directly to dehydration step without rinsing.
-
- Dehydrate in two changes of 100% ethyl alcohol. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
RESULTS:
| Acid epithelial mucin | Blue |
| Stromal mucin | Blue |
| Capsule of Cryptococcus neoformans | Blue |
| Collagen | Red |
| Muscle and cytoplasm | Yellow |
PROCEDURE NOTES:
-
- Acid clean all glassware/plasticware (Part 12086) and rinse thoroughly in several changes of distilled water.
- Drain slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during procedure.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps.
REFERENCES:
-
- Bancroft, John D., and Marilyn Gamble. Theory and Practice of Histological Techniques. 6th ed. Oxford: Churchill Livingstone Elsevier, 2008. 175-176.
- Carson, Freida L., and Christa Hladik Cappellano. Histotechnology: A Self-instructional Text. 4th ed. Chicago: ASCP Press, 2015. 151-153.
- Rekhtman, Natasha, and Justin Bishop. Quick Reference Handbook for Surgical Pathologists. Berlin: Springer, 2011. 69.
- Modifications developed by Newcomer Supply Laboratory.