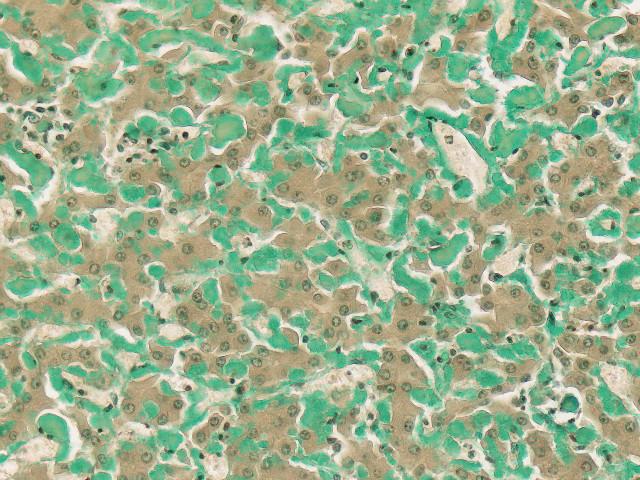
Sulfated Alcian Blue (SAB) Stain
Reagents for this procedure are sold as individual stain solutions and are available for purchase under separate part numbers with storage requirements and expiration date designated per bottle.
SOLUTIONS:
| 500 ml | 1 Liter | 1 Gallon | ||
| Alcian Blue Stain, Alcoholic | Part 1004A | |||
| Sodium Sulfate 1%, Aqueous | Part 1388A | |||
| Acetic Acid, Glacial, ACS | Part 10010A | |||
| Picric Acid, Saturated Alcoholic | Part 1337A | Part 1337B | Part 1337C | |
| Borax (Sodium Borate) 5%, Saturated Alcoholic | Part 1019A | Part 1019B | ||
| Hematoxylin Stain Set, Weigert Iron | Part 1409B | Part 1409A | ||
| Van Gieson Stain | Part 1404A | Part 1404B | ||
Additionally Needed:
| Alcohol, Ethyl Denatured, 95% | Part 10842 |
| Alcohol, Ethyl Denatured, 80% | Part 10843 |
| Xylene, ACS | Part 1445 |
| Alcohol, Ethyl Denatured, 100% | Part 10841 |
For storage requirements and expiration date refer to individual bottle labels.
APPLICATION:
Newcomer Supply Sulfated Alcian Blue (SAB) Stain assists in identifying the extraneous protein deposits of amyloidosis, specifically in myocardial and renal biopsy specimens.
METHOD:
Fixation: Formalin 10%, Phosphate Buffered (Part 1090)
Technique: Paraffin sections cut at 8-10 microns
Solutions: All solutions manufactured by Newcomer Supply Inc.
All Newcomer Supply stain procedures are designed to be used with Coplin jars filled to 40 ml following the staining procedure provided below.
STAINING PROCEDURE:
- Prepare the following three fresh solutions:
- SAB Staining Solution
Alcian Blue Stain, Alcoholic (Part 1004) 22 ml
Sodium Sulfate 1%, Aqueous (Part 1388) 22 ml
Acetic Acid, Glacial, ACS (Part 10010) 5 ml
Allow to stand for 30 minutes before use.
- Acetic Acid/Alcohol Solution
Alcohol, Ethyl Denatured, 95% 44 ml
Distilled Water 44 ml
Acetic Acid, Glacial, ACS 10 ml
- Picric Acid 2%, Alcoholic Working Solution
Alcohol, Ethyl Denatured, 80% 30 ml
Picric Acid, Saturated Alcoholic (Part 1337) 10 ml
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
- See Procedure Notes #1 and #2.
- Place in Acetic Acid/Alcohol Solution (Step #1b) for 2 minutes.
- Place slides in SAB Staining Solution (Step #1a) for 2 hours.
- Transfer directly to fresh Acetic Acid/Alcohol Solution (Step #1b) for 2 minutes.
- Wash well in tap water; rinse in distilled water.
- Alkalinize slides in Borax (Sodium Borate) 5%, Saturated Alcoholic (Part 1019) for 30 minutes.
- Wash well in tap water; rinse in distilled water.
- Prepare Working Hematoxylin Stain, Weigert Iron (Part 1409); combine and mix well.
- Solution A: Ferric Chloride, Aqueous 20 ml
- Solution B: Hematoxylin 1%, Alcoholic 20 ml
- Stain in Working Hematoxylin Stain, Weigert Iron for 5 minutes.
- Wash in tap water for 10 minutes; rinse in distilled water.
- Differentiate each slide individually; dip in Picric Acid 2%, Alcoholic Working Solution (Step #1c) for 20 seconds.
- Rinse briefly in tap water; 2-3 dips.
- Counterstain with Van Gieson Stain (Part 1404) for 3 minutes.
- Dehydrate quickly in two changes each of 95% and 100% ethyl alcohol. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
RESULTS:
| Amyloid deposits | Shades of green; pale to brilliant jade |
| Fibrin, muscle, cytoplasm | Yellow |
| Collagen, stroma | Red |
| Nuclei | Black |
Note: Green staining tissues other than amyloid are distinguishable on morphological grounds. Tissue mast cells stain a dense bright green, showing typical cytoplasmic granularity. Red blood cells occasionally appear very pale green; calcium deposits stain a dirty blue/green.
PROCEDURE NOTES:
- Drain staining rack/slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during staining procedure.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps.
REFERENCES:
- Lendrum, A. C., W. Slidders, and D. S. Fraser. “Renal Hyalin: A Study of Amyloidosis and Diabetic Fibrinous Vasculosis with New Staining Methods.” Journal of Clinical Pathology 25 (1972): 373-96.
- Pomerance, Ariela, Gerard Slavin, and Josephine McWatt. “Experience with the Sodium Sulphate-Alcian Blue Stain for Amyloid in Cardiac Pathology.” Journal of Clinical Pathology, 29 (1976): 22-26.
- Modifications developed by Newcomer Supply Laboratory.