Elastic, Skin
|
Validation Stain: Verhoeff
Other Applicable Stains: Gomori Aldehyde Fuchsin and Orcein
|
PRODUCT SPECIFICATIONS:
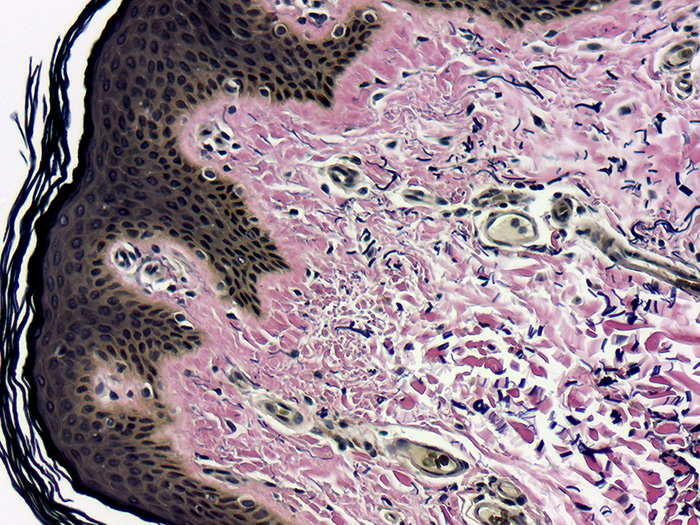
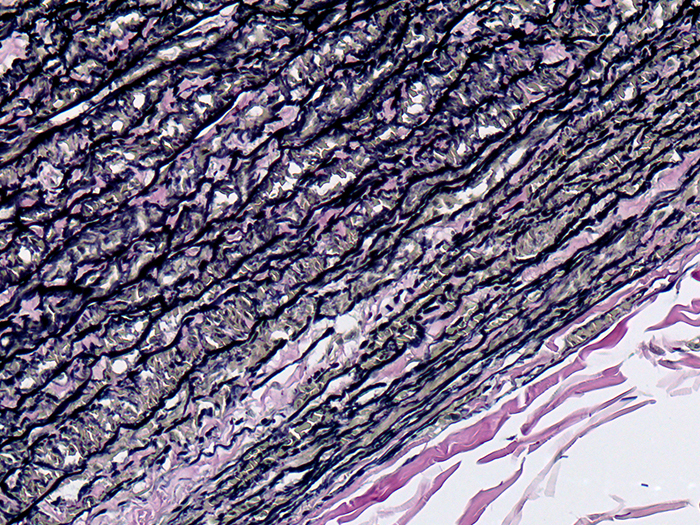
Tissue: Positive staining skin.
Fixation: Formalin 10%, Phosphate Buffered (Part 1090).
Section/Glass: Paraffin sections cut at 4 microns on Superfrost™ Plus slides.
Quality Control Stain: Verhoeff-Van Gieson Elastic quality control stained slide(s) included.
Reactivity: Guaranteed product specific reactivity for one year from date of receipt. Revalidate after one year to verify continued reactivity.
Storage: 15-30°C in a light deprived and humidity controlled environment.
Intended Use: To verify histological techniques and reagent reactivity.
Before using unstained control slides, review the enclosed stained slide(s) to ensure that this tissue source is acceptable for testing needs.
CONTROL SLIDE VALIDATION:
| With Elastic, Verhoeff Stain Kit: |
Part 9116A/B | Individual Stain Solution | |
| Solution A: |
Hematoxylin 5%, Alcoholic | 125/250 ml | Part 11623 |
| Solution B: |
Ferric Chloride 10%, Aqueous | 125/250 ml | Part 10856 |
| Solution C: |
Iodine, Lugol’s, Aqueous | 75/150 ml | Part 12092 |
| Solution D: |
Sodium Thiosulfate 5%, Aqueous | 250/500 ml | Part 1389 |
| Solution E: |
Van Gieson Stain | 250/500 ml | Part 1404 |
APPLICATION:
Newcomer Supply Elastic, Skin Control Slides are for the positive histochemical staining of elastic fibers in skin.
PRESTAINING PREPARATION:
-
- Heat dry sections in oven according to your laboratory protocol.
- Prepare fresh Verhoeff Working Solution by combining in the exact order listed, mixing well after each addition. Save for Step #5.
-
- Solution A: Hematoxylin 5%, Alcoholic 20 ml
- Solution B: Ferric Chloride 10%, Aqueous 8 ml
- Solution C: Iodine, Lugol’s, Aqueous 8 ml
-
- Prepare fresh Ferric Chloride 2%, Aqueous Solution for Step #7.
-
- Solution B: Ferric Chloride 10%, Aqueous 10 ml
- Distilled water 40 ml
-
STAINING PROCEDURE:
-
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
-
- See Procedure Notes #1 and #2.
-
- Stain in fresh Verhoeff Working Solution (Step #2) for 20 minutes.
-
- Discard solution after successful differentiation in Step #7.
-
- Rinse in several changes of tap water.
- Differentiate each slide individually in fresh Ferric Chloride 2%, Aqueous Solution (Step #3) with agitation; approximately 20 dips.
- Check differentiation: rinse well in tap water and check microscopically for black elastic staining with gray background.
-
- If necessary, return to Ferric Chloride 2%, Aqueous Solution until desired elastic differentiation is achieved.
- See Procedure Notes #3 and #4.
-
- Wash well in tap water.
- Place in Solution D: Sodium Thiosulfate 5%, Aqueous for 1 minute.
- Wash well in running tap water for 5 minutes.
- Counterstain in Solution E: Van Gieson Stain for 3 to 5 minutes.
- Dehydrate in two changes each of 95% and 100% ethyl alcohol. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
RESULTS:
| Elastic fibers/tissue/nuclei | Blue-black to black |
| Collagen | Red |
| Other tissue elements | Yellow |
PROCEDURE NOTES:
-
- Drain slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during procedure.
- If background is colorless, the section has been over-differentiated.
-
- Over-differentiated sections may be re-stained in Step #5 provided sections have not been treated with alcohol.
-
- Differentiation can vary between slides dependent upon the amount of elastic tissue present in sections.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps.
REFERENCES:
-
- Carson, Freida L., and Christa Hladik Cappellano. Histotechnology: A Self-instructional Text. 4th ed. Chicago: ASCP Press, 2015. 167-169.
- Mallory, Frank Burr, and James Homer Wright. Pathological Technique. 7th ed. Philadelphia, PA: W.B. Saunders Company, 1918. 118-119.
- Modifications developed by Newcomer Supply Laboratory.