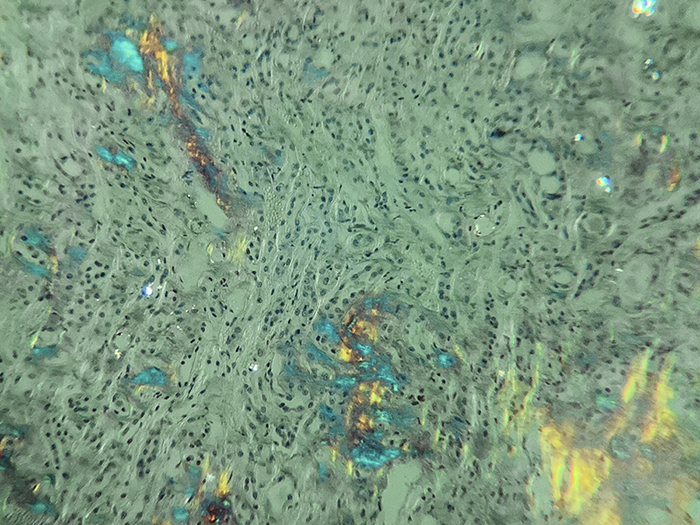
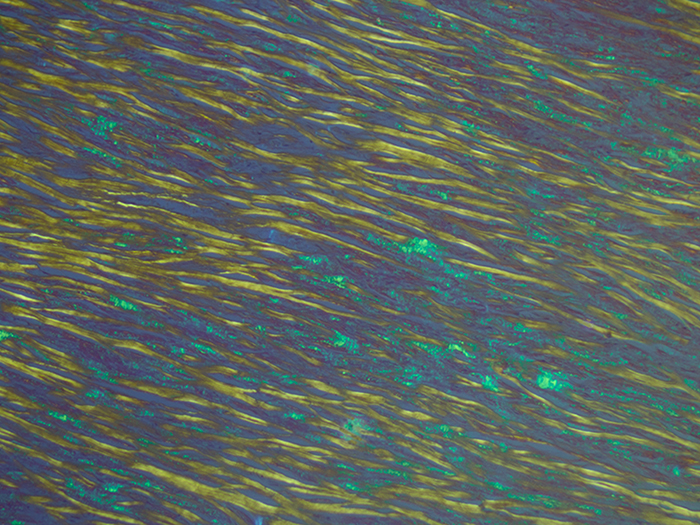
Amyloid, Animal
|
Validation Stain: Bennhold Congo Red
Other Applicable Stains: Puchtler Congo Red, Crystal Violet-Lieb and Thioflavin
|
-
- Due to the nature of the tissue the standard drying method for Amyloid Control Slides are oven dried
- Tissue sections are cut at 8 microns
PRODUCT SPECIFICATIONS:
Tissue: Positive staining animal organ.
Fixation: Formalin 10%, Phosphate Buffered (Part 1090).
Section/Glass: Paraffin sections cut at 8 microns on Superfrost™ Plus slides.
Quality Control Stain: Bennhold Congo Red quality control stained slide(s) included.
Reactivity: Guaranteed product specific reactivity for one year from date of receipt. Revalidate after one year to verify continued reactivity.
Storage: 15-30°C in a light deprived and humidity controlled environment.
Intended Use: To verify histological techniques and reagent reactivity.
Before using unstained control slides, review the enclosed stained slide(s) to ensure that this tissue source is acceptable for testing needs.
CONTROL SLIDE VALIDATION:
| With Amyloid, Bennhold Congo Red Stain Kit: | Part 9103A | Individual Stain Solution | |
| Solution A: | Congo Red Stain 1%, Aqueous | 250 ml | Part 1038 |
| Solution B: | Alkaline Alcohol | 250 ml | Part 1038 |
| Solution C: | Hematoxylin Stain, Mayer Modified | 250 ml | Part 1202 |
APPLICATION:
Newcomer Supply Amyloid, Animal Control Slides are for the positive histochemical staining of extraneous protein deposits in amyloidosis.
NEWCOMER SUPPLY VALIDATION PROCEDURE:
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
- See Procedure Notes #1 and #2.
- Place slides in Solution A: Congo Red Stain 1%, Aqueous for 1 hour.
Microwave Modification: See Procedure Note #3.
- Place slides in a plastic Coplin jar (Part 5184) containing Solution A: Congo Red Stain 1%, Aqueous and microwave at 70°C for 3 minutes.
- Rinse in two to three changes of tap water; rinse in distilled water.
- Differentiate in Solution B: Alkaline Alcohol, 5 to 30 seconds, agitating constantly until slide background is cleared of Solution A: Congo Red Stain 1%, Aqueous.
- Rinse in two to three changes of tap water; rinse in distilled water.
- Counterstain with Solution C: Hematoxylin Stain, Mayer Modified, 3 to 5 minutes, depending on preference of nuclear stain intensity.
- Wash in running tap water for 5 to 10 minutes.
- Dehydrate in two changes each of 95% and 100% ethyl alcohol. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
RESULTS:
| Light Field Microscopy: | ||
| Amyloid | Pink to red | |
| Nuclei | Blue | |
| Polarized Light: | ||
| Amyloid fluorescence | Apple green | |
PROCEDURE NOTES:
- Drain slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during procedure.
- The suggested microwave procedure has been tested at Newcomer Supply. This procedure is a guideline and techniques should be developed for use in your laboratory.
- For optimal results cut sections at 8-10 microns to provide more intense staining and allow smaller amyloid deposits to be identified. Thinner sections may show faint staining and sections thicker than 8-10 microns may display yellow birefringence.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps.
REFERENCES:
- Luna, Lee G. Histopathologic Methods and Color Atlas of Special Stains and Tissue Artifacts. Gaitheresburg, MD: American Histolabs, 1992. 366-367.
- Sheehan, Dezna C., and Barbara B. Hrapchak. Theory and Practice of Histotechnology. 2nd ed. St. Louis: Mosby, 1980. 177-178.
- Modifications developed by Newcomer Supply Laboratory.