Steiner-Steiner Modified Silver Stain Kit

![]()
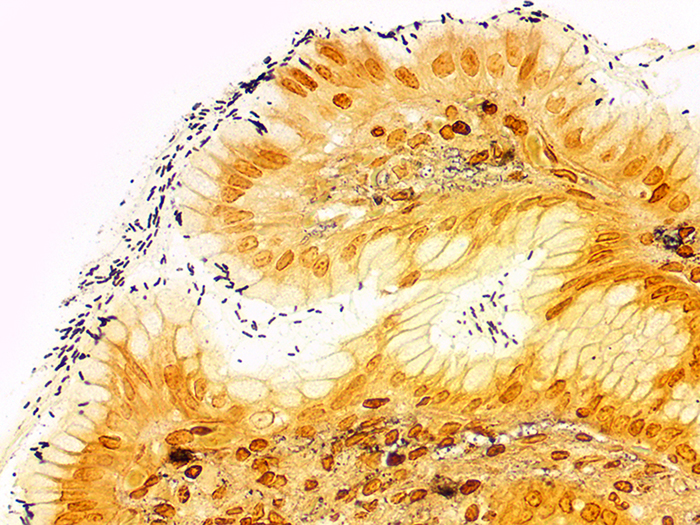
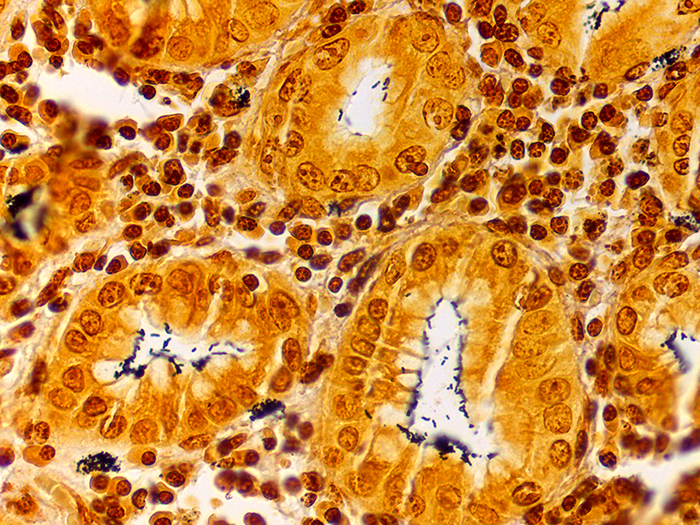
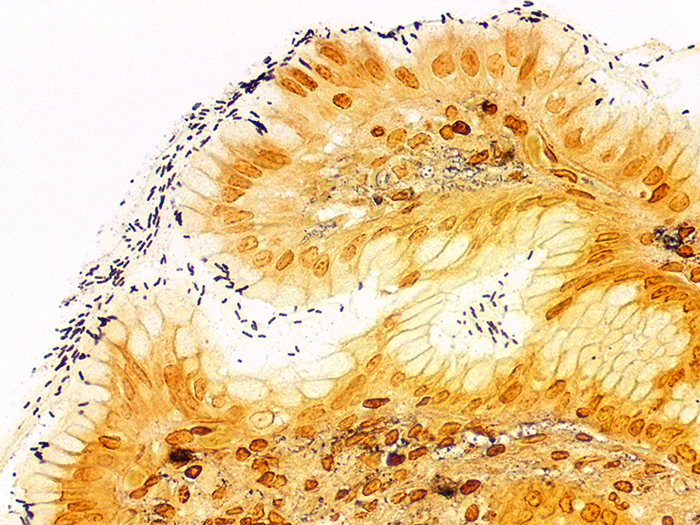
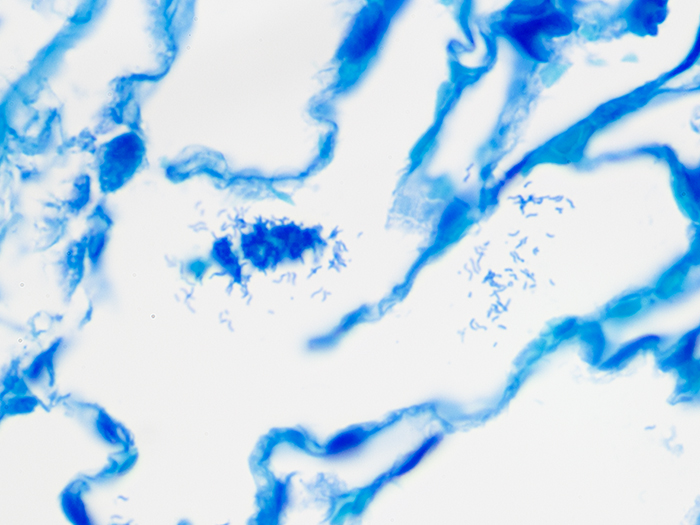
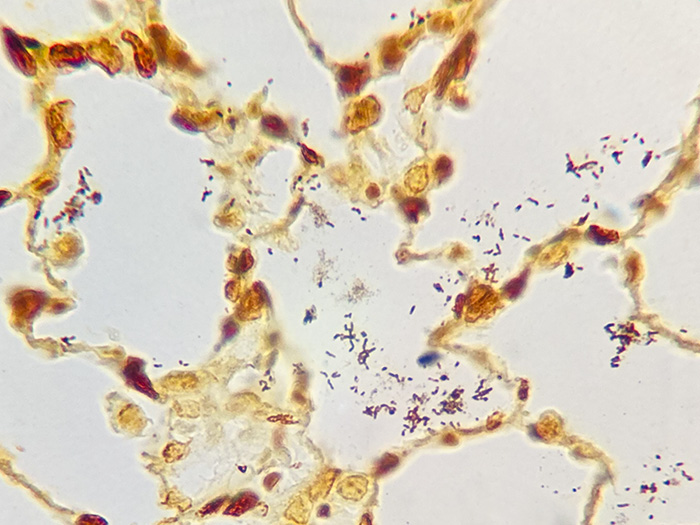
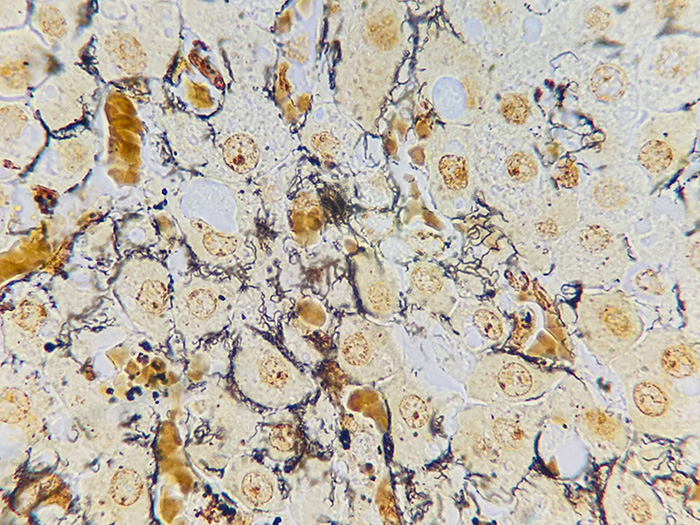
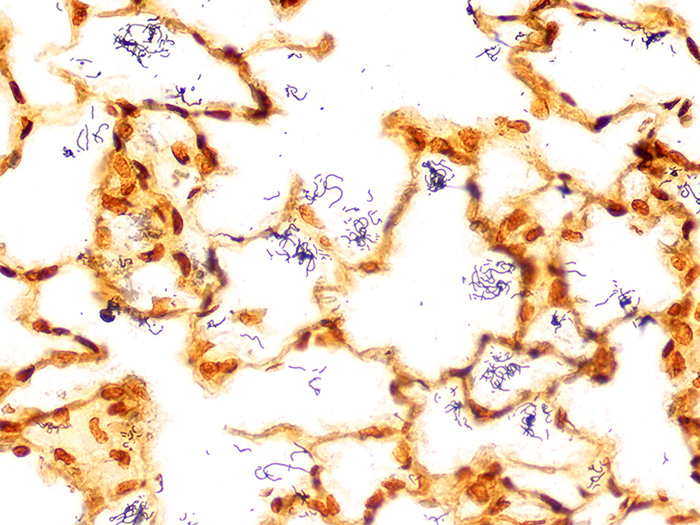
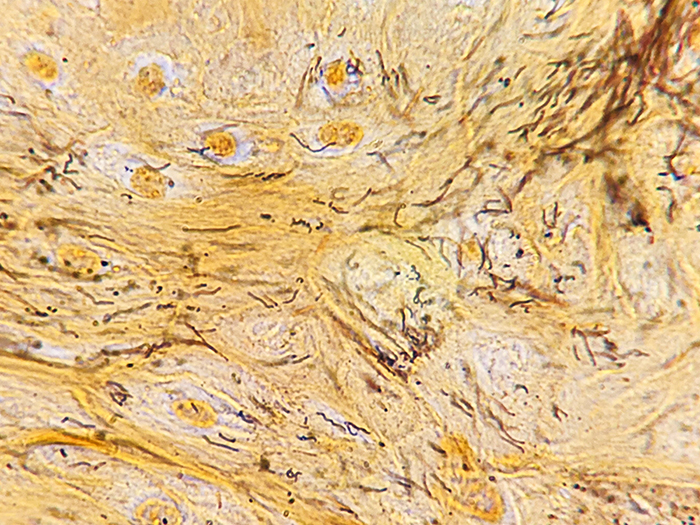
A silver stain for identifying spirochetes, Helicobacter pylori, Legionella pneumophila, other nonfilamentous bacteria and fungus.
- Faster, more efficient
- Microwave Modification Included
STEINER-STEINER MODIFIED SILVER STAIN KIT INCLUDES:
| Part 9171A | ||
| Solution A: | Uranyl Nitrate 1%, Aqueous | 250 ml |
| Solution B: | Silver Nitrate 1%, Aqueous | 250 ml |
| Solution C: | Gum Mastic 2.5%, Alcoholic | 175 ml x 2 |
| Ingredient D: | Hydroquinone, Powder | 5 grams |
| Mini Sampling Spoon |
COMPLIMENTARY POSITIVE CONTROL SLIDES: Enclosed are two complimentary unstained positive control slides for the initial verification of staining techniques and reagents. Verification must be documented by running one Newcomer Supply complimentary positive control slide along with your current positive control slide for the first run. Retain the second complimentary control slide for further troubleshooting, if needed.
Individual stain solutions and additional control slides may be available for purchase under separate part numbers at www.newcomersupply.com.
Additionally Needed:
| Hydrochloric Acid 5%, Aqueous | Part 12086 (for acid cleaning glassware) |
| Xylene, ACS | Part 1445 |
| Alcohol, Ethyl Denatured, 100% | Part 10841 |
| Alcohol, Ethyl Denatured, 95% | Part 10842 |
| Coplin Jar, Plastic | Part 5184 (for microwave modifications) |
For storage requirements and expiration date refer to individual bottle labels.
APPLICATION:
Newcomer Supply Steiner-Steiner Modified Silver Stain Kit procedure, with included microwave modifications, is a silver technique effective for the demonstration of spirochetes, Helicobacter pylori, Legionella pneumophila, other nonfilamentous bacteria and fungus.
METHOD:
Fixation: Formalin 10%, Phosphate Buffered (Part 1090)
Technique: Paraffin sections cut at 4 microns
Solutions: All solutions are manufactured by Newcomer Supply, Inc.
Newcomer Supply Stain Kits are designed to be used with Coplin jars filled to 40 ml following the provided staining procedure. Some solutions in the kit may contain extra volumes.
PRESTAINING PREPARATION:
-
- If necessary, heat dry tissue sections/slide in oven.
- All glassware/plasticware must be acid cleaned prior to use.
- See Procedure Notes #1 and #2.
- Preheat Solution A: Uranyl Nitrate 1%, Aqueous to 60°C in a water bath. Save for Step #9.
- Preheat Solution B: Silver Nitrate 1%, Aqueous to 60°C in a water bath. Save for Step #11.
- Prepare Hydroquinone Solution; combine and mix well.
- Ingredient D: Hydroquinone, Powder 5 gm (or one rounded scoop with reusable mini sampling spoon)
- Distilled Water 25 ml
-
- Prepare fresh Reducing Solution by combining in order listed.
- Hydroquinone Solution (Step #5) 25 ml
- Solution C: Gum Mastic 2.5%, Alcoholic 15 ml
- Solution B: Silver Nitrate 1%, Aqueous 6 ml
- Solution will turn milky white after addition of Gum Mastic.
- Preheat solution in 45°C water bath. Save for Step #15.
- Do not preheat solutions if using Microwave Modifications.
- Prepare fresh Reducing Solution by combining in order listed.
STAINING PROCEDURE:
-
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
-
-
- See Procedure Note #3
-
-
- Sensitize in preheated Solution A: Uranyl Nitrate 1%, Aqueous (Step #3) for 10 minutes in a 60°C water bath.
Microwave Modification: See Procedure Note #4.
-
-
-
- Place slides in a plastic Coplin jar with Solution A: Uranyl Nitrate 1%, Aqueous. Microwave at 70°C for 1 minute.
-
-
-
- Rinse well in several changes of distilled water.
- Place slides in preheated Solution B: Silver Nitrate 1%, Aqueous (Step #4) and incubate in a 60°C water bath for 15 minutes.
Microwave Modification:
-
-
- Place slides in a plastic Coplin jar with Solution B: Silver Nitrate 1%, Aqueous. Microwave at 70°C for 1 minute.
- Remove from microwave, cover and let sit for 1 minute.
-
-
- Rinse well in several changes of distilled water.
- Excessive rinsing may cause nucleus to pick up silver.
- Dip 5 times in two changes each of 95% and 100% ethyl alcohols.
- Place in Solution C: Gum Mastic 2.5%, Alcoholic for 3 minutes.
- Place slides in preheated Reducing Solution (Step #6) in 45°C water bath for 10-30 minutes with frequent agitation. Examine microscopically after 10 minutes of incubation.
- Check microscopically by dipping slide in 100% alcohol.
- Review for desired staining results.
- If necessary, return to warm solution; check every 2-5 minutes until desired results are achieved.
Microwave Modification:
-
- Place slides in a plastic Coplin jar with Reducing Solution. Microwave at 70°C for 1 minute. Remove from microwave.
- Pipette solution twice with plastic pipette to evenly distribute heated solution.
- Cover and let sit for 1 minute.
- Check microscopically by dipping slide in 100% alcohol.
- Review for desired staining results.
- If necessary, return to warm solution, check every 1 minute until desired results are achieved.
- Rinse well in several changes of distilled water.
-
- Directly dehydrate in two changes of 100% ethyl alcohol. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
RESULTS:
| Spirochetes | Dark brown to black |
| Helicobacter pylori | Dark brown to black |
| Legionella pneumophila | Dark brown to black |
| Nonfilamentous bacteria and fungus | Dark brown to black |
| Background | Golden brown |
PROCEDURE NOTES:
-
- Acid clean all glassware/plasticware (Part 12086) and rinse thoroughly in several changes of distilled water.
- Plastic (Part 5500), plastic-tipped or paraffin coated metal forceps must be used with any silver solution to prevent precipitation of silver salts. No metals of any kind should be in contact with any silver solution. Only glass thermometers should be used.
- Drain slides after each step to prevent solution carry over.
- The suggested microwave procedure has been tested at Newcomer Supply. This procedure is a guideline and techniques should be developed for your laboratory.
- If using a xylene substitute, follow manufacturer’s recommendation for deparaffinization and clearing steps.
REFERENCES:
-
- Garvey, Winsome. “Some Favorite Silver Stains.” The Journal of Histotechnology 3 (1996): 269-278.
- Luna, Lee G. Histopathologic Methods and Color Atlas of Special Stains and Tissue Artifacts. Gaitheresburg, MD: American Histolabs, 1992. 218-219.
- Steiner, Gabriel, and Grete Steiner. “New Simple Silver Stain for Demonstration of Bacteria, Spirochetes and Fungi in Sections of Paraffin Embedded Tissue Blocks.” Journal of Laboratory Clinical Medicine 29 (1944). 868-871.
- Swisher, Billie. “Modified Steiner Procedure for Microwave Staining of Spirochetes and Nonfilamentous Bacteria.” The Journal of Histotechnology4 (1987): 241-243.
- Modifications developed by Newcomer Supply Laboratory.