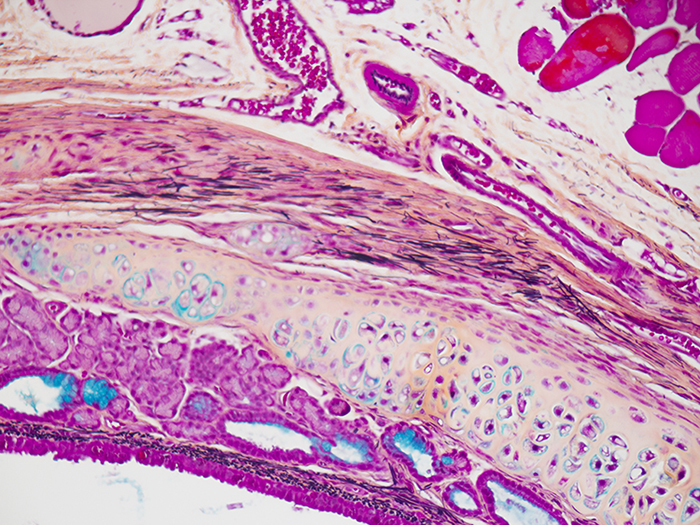
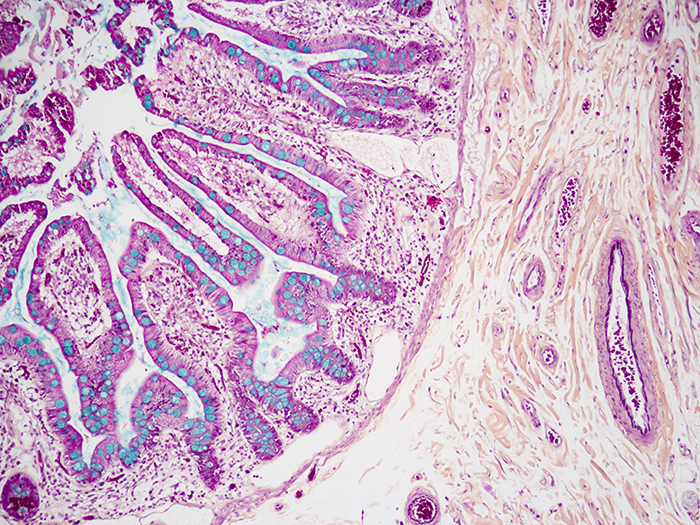
Movat-Russell Modified Pentachrome Stain Kit
Stains five connective tissue elements; mucin, fibrin, elastic fibers, muscle, and collagen, along with nuclei.
MOVAT-RUSSELL MODIFIED PENTACHROME STAIN KIT INCLUDES:
| Part 9150A | ||
| Solution A: | Alcian Blue Stain 1%, Aqueous | 250 ml |
| Solution B: | Ammonium Hydroxide 28-30%, ACS | 50 ml |
| Solution C: | Hematoxylin 10%, Alcoholic | 100 ml |
| Solution D: | Ferric Chloride 10%, Aqueous | 100 ml |
| Solution E: | Iodine, Verhoeff, Aqueous | 100 ml |
| Solution F: | Ferric Chloride 2%, Aqueous | 250 ml |
| Solution G: | Sodium Thiosulfate 5%, Aqueous | 250 ml |
| Solution H: | Crocein Scarlet 7B Stain, Aqueous | 250 ml |
| Solution I: | Acid Fuchsin Stain, Aqueous | 100 ml |
| Solution J: | Phosphotungstic Acid 5%, Aqueous | 500 ml |
| Solution K: | Orange G Stain 1%, Aqueous | 250 ml |
| Solution L: | Acetic Acid 0.5%, Aqueous | 500 ml |
COMPLIMENTARY POSITIVE CONTROL SLIDES: Enclosed are two complimentary unstained positive control slides for the initial verification of staining techniques and reagents. Verification must be documented by running one Newcomer Supply complimentary positive control slide along with your current positive control slide for the first run. Retain the second complimentary control slide for further troubleshooting, if needed.
Individual stain solutions and additional control slides may be available for purchase under separate part numbers.
Additionally Needed:
| Xylene, ACS | Part 1445 |
| Alcohol, Ethyl Denatured, 100% | Part 10841 |
| Alcohol, Ethyl Denatured, 95% | Part 10842 |
For storage requirements and expiration date refer to individual bottle labels.
APPLICATION:
Newcomer Supply Movat-Russell Modified Pentachrome Stain Kit provides a single staining procedure that demonstrates five connective tissue elements; mucin, fibrin, elastic fibers, muscle, and collagen.
METHOD:
Fixation: Formalin 10%, Phosphate Buffered (Part 1090)
Technique: Paraffin sections cut at 4 microns
Solutions: All solutions are manufactured by Newcomer Supply, Inc.
All Newcomer Supply Stain Kits are designed to be used with Coplin jars filled to 40 ml following the staining procedure provided below. Some solutions in the kit may contain extra volumes.
STAINING PROCEDURE:
-
- If necessary, heat dry tissue sections/slides in oven.
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
-
- See Procedure Notes #1 and #2.
-
- Stain in Solution A: Alcian Blue Stain 1%, Aqueous for 20 minutes.
- Wash in running tap water for 5 minutes.
- Prepare fresh Alkaline Alcohol Solution; combine and mix well.
-
- Solution B: Ammonium Hydroxide 28-30%, ACS 5 ml
- Alcohol, Ethyl Denatured, 95% (Part 10842) 45 ml
-
- Place slides in fresh Alkaline Alcohol Solution for 30 minutes.
- Wash in running tap water for 10 minutes; rinse in distilled water.
-
- See Procedure Note #3.
-
- Prepare fresh Hematoxylin Working Stain Solution just before use in the order given; combine and mix well.
-
- Solution C: Hematoxylin 10%, Alcoholic 10 ml
- Alcohol, Ethyl Denatured, 100% (Part 10841) 10 ml
- Solution D: Ferric Chloride 10%, Aqueous 10 ml
- Solution E: Iodine, Verhoeff, Aqueous 10 ml
-
- Stain in fresh Hematoxylin Working Stain Solution for 15 minutes.
-
- Discard after successful differentiation in Step #11.
-
- Rinse in several changes of distilled water.
- Differentiate one slide at a time in Solution F: Ferric Chloride 2%, Aqueous until elastic fibers contrast sharply with background; approximately 5-10 dips.
-
- See Procedure Note #4.
-
- Rinse in distilled water.
- Place in Solution G: Sodium Thiosulfate 5%, Aqueous for 1 minute.
- Wash in running tap water for 5 minutes; rinse in distilled water.
- Prepare Crocein Scarlet-Acid Fuchsin Solution:
-
- Solution H: Crocein Scarlet 7B Stain, Aqueous 40 ml
- Solution I: Acid Fuchsin Stain, Aqueous 10 ml
-
- Stain in Crocein Scarlet-Acid Fuchsin Solution for 1 minute.
- Rinse in several changes of distilled water.
- Rinse in Solution L: Acetic Acid 0.5%, Aqueous for 30 seconds.
- Place slides in Solution J: Phosphotungstic Acid 5%, Aqueous; two changes of 5 minutes each.
- Rinse in Solution L: Acetic Acid 0.5%, Aqueous.
- Stain in Solution K: Orange G Stain 1%, Aqueous for 15 minutes.
- Dehydrate through three changes of 100% ethyl alcohol, 10 dips each. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
-
- Do not use 95% ethyl alcohol in the dehydration step.
-
RESULTS:
| Nuclei and elastic fibers | Black |
| Collagen and reticular fibers | Yellow |
| Ground substance and mucin | Blue |
| Fibrinoid, fibrin | Intense red |
| Muscle | Red |
PROCEDURE NOTES:
-
- Drain slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during procedure.
- Completely remove Alkaline Alcohol Solution with running tap water. Failure to do so will inhibit the subsequent staining steps.
- Do not over-differentiate in Solution F. If background is colorless, the section may be over-differentiated. Over-differentiated sections can be restained in Hematoxylin Working Stain Solution (Step #9) if sections have not been treated with an alcohol/dehydration step.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps.
REFERENCES:
-
- Carson, Freida L., and Christa Hladik. Histotechnology: A Self-Instructional Text. 3rd ed. Chicago, Ill.: American Society of Clinical Pathologists, 2009. 172-174.
- Movat, Henry, “Demonstration of All Connective Tissue Elements in a Single Section”. AMA Archives of Pathology. 1955; 60 (3): 289–295.
- Russell H. K. Jr. “A Modification of Movat’s Pentachrome Stain”. AMA Archives of Pathology.1972; 94 (2): 187–191.
- Modifications developed by Newcomer Supply Laboratory.