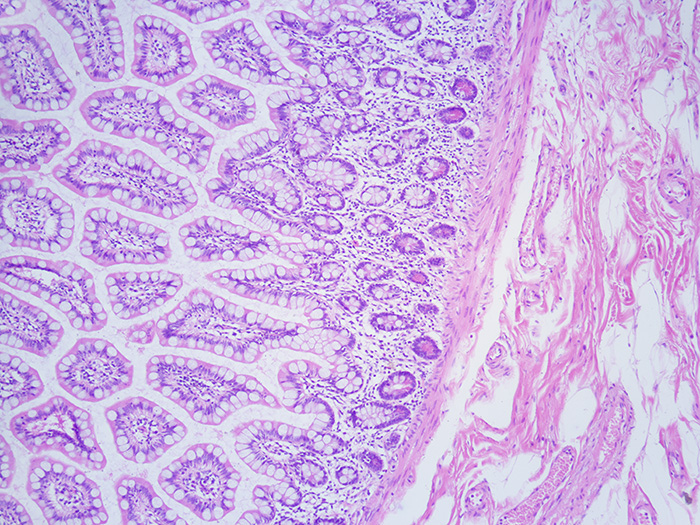
Hematoxylin and Eosin (H&E) Progressive Stain
Reagents for this procedure are sold as individual stain solutions and are available for purchase under separate part numbers with storage requirements and expiration date designated per bottle.
SOLUTIONS:
| 500 ml | 1 Liter | 1 Gallon | |
| Hematoxylin Stain, Mayer Modified | Part 1202A | Part 1202B | Part 1202C |
| Eosin Y Working Solution | Part 1072A | Part 1072B | Part 1072C |
Additionally Needed For H&E Staining:
| Hematoxylin & Eosin (H&E) Control Slides | Part 4278 |
| Xylene, ACS | Part 1445 |
| Alcohol, Ethyl Denatured, 100% | Part 10841 |
| Alcohol, Ethyl Denatured, 95% | Part 10842 |
| Lithium Carbonate, Saturated Aqueous OR Scott Tap Water Substitute |
Part 12215 OR Part 1380 |
| Alcohol, Ethyl Denatured, 70% | Part 10844 |
For storage requirements and expiration date refer to individual product labels.
APPLICATION:
Newcomer Supply Hematoxylin & Eosin (H&E) Progressive Stain is used for screening specimens in anatomic pathology, as well as for research, smears, touch preps and other applications. In progressive staining, tissue is left in the hematoxylin solution just long enough to reach the proper endpoint. It may be necessary to examine the slides at different timed intervals to determine when staining is optimal.
Hematoxylin Stain, Mayer Modified is a ready-to-use progressive hematoxylin that does not require filtering and does not contain chloral hydrate or alcohol. Due to the progressive staining nature of Mayer hematoxylin, over-staining is less likely and an acid alcohol differentiation step is not required in the staining process.
Eosin Y Working Solution is a ready-to-use counterstain with the ability to distinguish between the cytoplasm of different types of cells by staining cytoplasmic components differing shades and intensities of pink to red.
Quality Control: Since hematoxylin and eosin staining is the foundation of the diagnostic process, maintaining quality is of critical importance. Change staining solutions on a regular basis according to laboratory protocol. Procedures will vary between laboratories depending upon volume of slides, automation vs manual staining, chemical hygiene and solution integrity. The longevity of hematoxylin and eosin depend upon these factors and stain quality should be regularly screened with the use of an H&E control slide.
METHOD:
Fixation: Formalin 10%, Phosphate Buffered (Part 1090)
Technique: Paraffin sections cut at 5 microns
Solutions: All solutions are manufactured by Newcomer Supply, Inc.
STAINING PROCEDURE:
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
- See Procedure Notes #1 and #2.
- Stain with Hematoxylin Stain, Mayer Modified, 10-20 minutes, depending on preference of nuclear stain intensity.
- Wash well in tap water for 3 minutes.
- Blue slides in Lithium Carbonate, Saturated Aqueous (Part 12215) or Scott Tap Water Substitute (Part 1380) for 10 dips.
- See Procedure Note #3.
- Wash in three changes of tap water; rinse in distilled water.
- Drain excess water from rack/slides; proceed to 70% alcohol for 10 dips.
- Counterstain in Eosin Y Working Solution for 30 seconds to 3 minutes, depending on preference of intensity.
- Dehydrate in two changes of 95% ethyl alcohol for 1 minute each and two changes of 100% ethyl alcohol, 10 dips each. Clear in three changes of xylene, 10 dips each; coverslip with compatible mounting medium.
RESULTS:
| Nuclei | Blue |
| Erythrocytes and eosinophilic granules | Bright pink to red |
| Cytoplasm and other tissue elements | Various shades of pink |
PROCEDURE NOTES:
- Drain staining rack/slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during staining procedure.
- Some laboratory tap water may be alkaline enough to accomplish satisfactory bluing, allowing the elimination of Step #4.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps.
REFERENCES:
- Carson, Frieda, Histotechnology: A Self-Instructional Text. 3rd ed. Chicago: ASCP Press, 2009. 114-115.
- Luna, Lee G. Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology. 3rd ed. New York: Blakiston Division, McGraw-Hill, 1968. 36-38.
- Modifications developed by Newcomer Supply Laboratory.