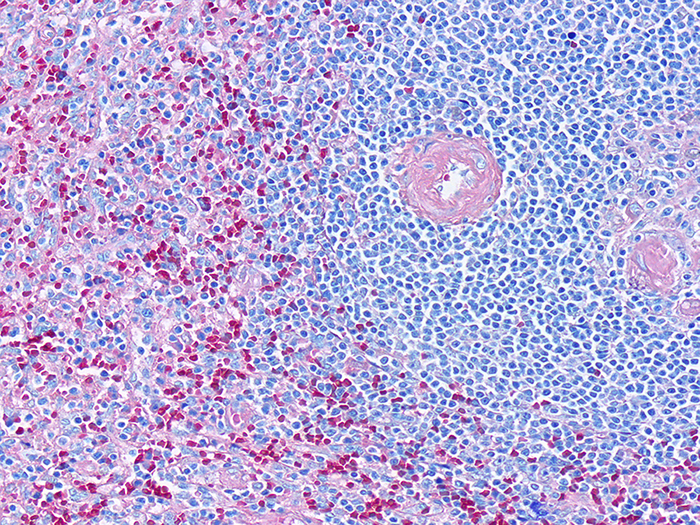
Giemsa, May-Grunwald
Reagents for this procedure are sold as individual stain solutions and are available for purchase under separate part numbers with storage requirements and expiration date designated per bottle.
Tech Memo 1: May-Grunwald Giemsa Stain for Paraffin Sections
SOLUTIONS:
| 500 ml | 1 Liter | |
| Jenner Stock Stain | Part 1210A | Part 1210B |
| Giemsa Stock Stain, Wolbach | Part 1121A | Part 1121B |
Additionally Needed:
| Giemsa Control Slides | Part 4240 |
| Xylene, ACS | Part 1445 |
| Alcohol, Ethyl Denatured, 100% | Part 10841 |
| Alcohol, Ethyl Denatured, 95% | Part 10842 |
| Alcohol, Methanol Anhydrous, ACS | Part 12236 |
| Acetic Acid 1%, Aqueous | Part 10012 |
For storage requirements and expiration date refer to individual product labels.
APPLICATION:
Newcomer Supply May-Grunwald Giemsa (MGG) Stain procedure for paraffin sections, is used for differential staining of hematopoietic tissue as well as demonstration of some microorganisms.
May-Grunwald refers to a combined stain of eosin and methylene blue; the Jenner Stock Stain. Giemsa refers to a mixture of methylene blue, eosin and Azure B; the Giemsa Stock Stain, Wolbach. The use of Jenner Stock Stain and Giemsa Stock Stain, Wolbach, provides intense colorization results in the MGG Stain.
METHOD:
Fixation: According to protocol for hematopoietic tissue
- See Procedure Note #1.
Technique: Paraffin sections cut at 5 microns
Solutions: All solutions are manufactured by Newcomer Supply, Inc.
All Newcomer Supply stain procedures are designed to be used with Coplin jars filled to 40 ml following the staining procedure provided below.
STAINING PROCEDURE:
- Deparaffinize sections thoroughly in three changes of xylene, 3 minutes each. Hydrate through two changes each of 100% and 95% ethyl alcohols, 10 dips each. Wash well with distilled water.
- See Procedure Notes #2 and #3.
- Rinse in two changes of Methanol (Part 12236); 3 minutes each.
- Prepare fresh Working Jenner Stain just prior to use; combine and mix well.
- Jenner Stock Stain 20 ml
- Distilled Water 20 ml
- Place slides in fresh Working Jenner Stain for 6 minutes.
- Prepare fresh Working Giemsa Stain just prior to use; combine and mix well.
- Giemsa Stock Stain, Wolbach 3 ml
- Distilled Water 47 ml
- Place slides, without rinsing, directly from Working Jenner Stain into fresh Working Giemsa Stain for 45 minutes.
- Rinse quickly in distilled water.
- Differentiate each slide individually in Acetic Acid 1%, Aqueous (Part 10012); 6-10 dips.
- Check microscopically for well differentiated nuclei.
- Rinse in distilled water.
- Dehydrate in two changes each of 95% and 100% ethyl alcohol. Clear in three changes of xylene, 10 dips each; coverslip with
RESULTS:
| Nuclei | Blue/violet |
| Cytoplasm | Pink/rose to lighter blue shades |
| Bacteria | Blue |
PROCEDURE NOTES:
- Zenker Fixative, Modified, Zinc Chloride (Part 1461) and B-5 Fixative Modified, Zinc Chloride (Part 1015) are the preferred fixatives for hematopoietic tissue; Formalin 10%, Phosphate Buffered (Part 1090) or other well fixed tissue is acceptable.
- Drain staining rack/slides after each step to prevent solution carry over.
- Do not allow sections to dry out at any point during staining procedure.
- The color range of the stained cells may vary depending upon fixation and degree of differentiation.
- If using a xylene substitute, closely follow the manufacturer’s recommendations for deparaffinization and clearing steps.
REFERENCES:
- Carson, Freida L., and Christa Hladik Cappellano. Histotechnology: A Self-instructional Text. 4th ed. Chicago: ASCP Press, 2015. 130-131.
- Luna, Lee G. Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology. 3rd ed. New York: Blakiston Division, McGraw-Hill, 1968. 121-122.
- Shapiro, Stanley H., and Hilda Laufer. “Observations on Fixation and Staining of Bone Marrow Biopsies.” The Journal of Histotechnology 11.3 (1988): 145-47.
- Sheehan, Dezna C., and Barbara B. Hrapchak. Theory and Practice of Histotechnology. 2nd ed. St. Louis: Mosby, 1980. 157.
- Modifications developed by Newcomer Supply Laboratory.
Tech Memo 2: May-Grunwald Giemsa Stain for Smears
SOLUTIONS:
| 500 ml | 1 Liter | |
| Jenner Stock Stain | Part 1210A | Part 1210B |
| Giemsa Stock Stain, Wolbach | Part 1121A | Part 1121B |
Additionally Needed:
| Alcohol, Methanol Anhydrous, ACS | Part 12236 |
For storage requirements and expiration date refer to individual bottle labels.
APPLICATION:
Newcomer Supply May-Grunwald Giemsa (MGG) Stain procedure for smears, is used for differential staining and morphological inspection of peripheral blood smears and bone marrow smears/films.
May-Grunwald refers to a combined stain of eosin and methylene blue; the Jenner Stock Stain. Giemsa refers to a mixture of methylene blue, eosin and Azure B; the Giemsa Stock Stain, Wolbach. The use of the Jenner Stock Stain and Giemsa Stock Stain, Wolbach provides intense colorization results in the MGG Stain.
METHOD:
Technique: Coplin jar or flat staining rack method
Solutions: All solutions are manufactured by Newcomer Supply, Inc.
STAINING PROCEDURE:
- Prepare within an accepted time frame, a well-made blood smear or bone marrow smear/film per your laboratories protocol, with a focus on uniform cell distribution.
- Allow smear(s) to thoroughly air-dry prior to staining.
- Fix smears by flooding slides with Methanol (Part 12236) for 10-30 seconds.
- Prepare fresh Working Jenner Stain just prior to use; combine and mix well.
- Jenner Stock Stain 20 ml
- Distilled Water 20 ml
- Or prepare lesser volumes as needed.
- Place slides in fresh Working Jenner Stain for 5-6 minutes.
- Prepare fresh Working Giemsa Stain just prior to use; combine and mix well.
- Giemsa Stock Stain, Wolbach 5 ml
- Distilled Water 45 ml
- Or prepare lesser volumes as needed.
- Place slides, without rinsing, directly from Working Jenner Stain into fresh Working Giemsa Stain for 30 minutes.
- Rinse smears well in distilled water to completely differentiate.
- Air-dry slides in a vertical position; examine microscopically.
RESULTS:
| Erythrocytes | Light pink to light purple |
| Platelets | Granules – Reddish purple |
| Lymphocytes/monocytes | Nuclei – Dark purple |
| Cytoplasm – Sky blue | |
| Neutrophils | Nuclei – Dark blue |
| Granules – Reddish purple | |
| Cytoplasm – Pale pink | |
| Eosinophils | Nuclei – Blue |
| Granules – Red/orange red | |
| Cytoplasm – Blue | |
| Basophils | Nuclei – Dark blue |
| Granules – Purple |
PROCEDURE NOTES:
- The timings provided in this procedure are suggested ranges. Optimal staining times will depend upon staining intensity preference.
- Drain slides after each step to prevent solution carry over.
REFERENCES:
- Carson, Freida L., and Christa Hladik Cappellano. Histotechnology: A Self-instructional Text. 4th ed. Chicago: ASCP Press, 2015. 130-131.
- McPherson, Richard and Matthew Pincus. Henry’s Clinical Diagnosis and Management by Laboratory Methods. 22nd ed. Philadelphia: Elsevier Saunders, 2011. 522-531.
- Sheehan, Dezna C., and Barbara B. Hrapchak. Theory and Practice of Histotechnology. 2nd ed. St. Louis: Mosby, 1980. 157.
- Modifications developed by Newcomer Supply Laboratory.