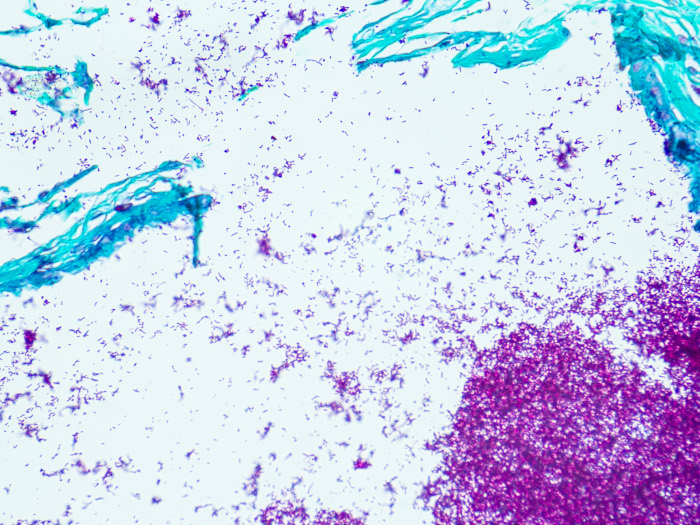
Fite, Nocardia, Artificial
|
Validation Stain: AFB-Fite
|
Procedural control for Nocardia sp. and leprosy
PRODUCT SPECIFICATIONS:
Tissue: Positive staining rat lung.
Fixation: Formalin 10%, Phosphate Buffered (Part 1090).
Section/Glass: Paraffin sections cut at 4 microns on Superfrost™ Plus slides.
Quality Control Stain: AFB, Fite quality control stained slide(s) included.
Reactivity: Guaranteed product specific reactivity for one year from date of receipt. Revalidate after one year to verify continued reactivity.
Storage: 15-30°C in a light deprived and humidity controlled environment.
Intended Use: To verify histological techniques and reagent reactivity.
Before using unstained control slides, review the enclosed stained slide(s) to ensure that this tissue source is acceptable for testing needs.
CONTROL SLIDE VALIDATION:
| With AFB, Fite Stain Kit: | Part 91013A | Individual Stain Solution | |
| Solution A: | Xylene/Peanut Oil, 2:1 | 500 ml | Part 1449 |
| Solution B: | Carbol Fuchsin Stain, Ziehl-Neelsen | 250 ml | Part 1030 |
| Solution C: | Acid Alcohol 1% | 250 ml | Part 10011 |
| Solution D: | Light Green SF Yellowish 0.1%, Aqueous | 250 ml | Part 12203 |
APPLICATION:
Newcomer Supply Fite, Nocardia, Artificial Control Slides are for the positive histochemical staining of Nocardia sp. and acid fast bacilli in tissue sections. Nocardia asteroides, purchased from Remel Microbiology Products, is used to produce the positive control tissue.
PRESTAINING PREPARATION:
-
- Heat dry sections in oven according to your laboratory protocol.
- Filter Solution B: Carbol Fuchsin Stain, Ziehl-Neelsen with high quality filter paper.
- Prepare Diluted Acid Alcohol Solution:
-
- Solution C: Acid Alcohol 1% 20 ml
- Distilled Water 20 ml
-
NEWCOMER SUPPLY VALIDATION PROCEDURE:
-
- Deparaffinize slides in Solution A: Xylene/Peanut Oil, 2:1, two changes for 12 minutes each.
-
- See Procedure Note #1.
-
- Drain slides, wipe off excess oil, and blot to opacity taking care to remove residual oil.
-
- See Procedure Note #2.
-
- Stain in freshly filtered Solution B: Carbol Fuchsin Stain, Ziehl-Neelsen for 15 minutes at room temperature.
- Rinse well in distilled water.
- Differentiate slides individually in Diluted Acid Alcohol Solution (Step #3) until background is pale pink; 10-20 dips.
- Quickly rinse in distilled water and check microscopically for correct differentiation.
- Rinse well in distilled water.
- Counterstain in Solution D: Light Green SF Yellowish 0.1%, Aqueous; 5-10 dips.
- Rinse in distilled water.
- Blot excess water from slide and air-dry or oven-dry completely.
- Dip dried slides in xylene and coverslip with a compatible mounting medium.
- Deparaffinize slides in Solution A: Xylene/Peanut Oil, 2:1, two changes for 12 minutes each.
RESULTS:
| Nocardia | Red |
| Acid-fast bacilli | Red |
| Other tissue elements | Green |
PROCEDURE NOTES:
-
- Acid-fastness of the organisms is enhanced when the waxy capsule is protected by the mixture of xylene/peanut oil and the avoidance of dehydrating solutions.
- It is important to blot well, residual oil may produce staining artifact.
- A small percentage of Nocardia sp. organisms may resist taking the red stain and remain green due to the growth phase of the individual organism.
- If using a xylene substitute, follow manufacturer’s recommendation for coverslipping step.
REFERENCES:
-
- Carson, Freida L., and Christa Cappellano. Histotechnology: A Self-instructional Text. 5th ed. Chicago: ASCP Press, 2020. 215-216.
- Fite, George, P.J. Cambre and M.H. Turner. “Procedure for Demonstrating Lepra Bacilli in Paraffin Sections”. Archives of Pathology 43 (1947). 624-625.
- Sheehan, Dezna C., and Barbara B. Hrapchak. Theory and Practice of Histotechnology. 2nd ed. St. Louis: Mosby, 1980. 237.
- Modifications developed by Newcomer Supply Laboratory.